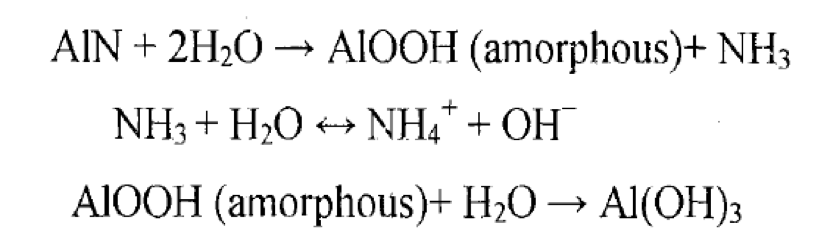AlN ceramics exhibit excellent insulation, high thermal conductivity, superior high-temperature resistance, corrosion resistance, and a thermal expansion coefficient that matches silicon, making them an ideal heat dissipation and packaging material for new-generation large-scale integrated circuits, semiconductor module circuits, and high-power devices. However, AlN powder—the raw material for producing high temperature AlN ceramics—is highly susceptible to hydrolysis. This not only complicates its transportation and storage but, more critically, reduces its nitrogen content after hydrolysis, significantly degrading the performance of the final AlN ceramics. Additionally, the hydrolytic instability of AlN powder hinders the development of water-based forming processes for industrial AlN ceramic products.

Thus, the hydrolytic sensitivity of AlN powder has become the primary obstacle to its widespread application. Overcoming this issue and improving the powder’s hydrolysis resistance to make it suitable for water-based wet forming has become a key research focus in the field of AlN ceramics in recent years.
Hydrolysis Mechanism
The conditions and products of AlN powder hydrolysis vary slightly among different studies. Some researchers suggest that the powder first reacts with water to form amorphous AlOOH and ammonia gas. The ammonia then reacts with water to produce OH⁻, increasing the solution's pH. Under certain temperature and acidity conditions, AlOOH further reacts with water to form Al(OH)₃.
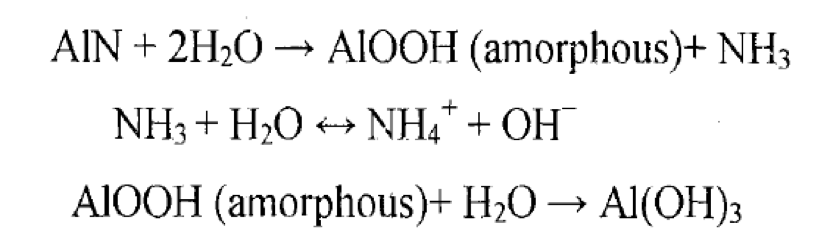
How to Determine if AlN Has Hydrolyzed?
Since hydrolysis generates ammonia gas, which ionizes into NH₄⁺ and OH⁻ in water, the pH of the solution changes, making pH a key indicator of hydrolysis extent.
Additionally, XRD can be used for phase analysis before and after hydrolysis to qualitatively identify new phases and assess hydrolysis products/degree. SEM reveals morphological changes in AlN particles, providing qualitative insights into hydrolysis progression. TEM, beyond morphology analysis,can examine the crystal structure of hydrolysis products. Since hydrolysis consumes AlN powder (via ammonia release), measuring the powder’s mass loss before and after hydrolysis is another critical metric.
How to Mitigate AlN Hydrolysis?
Hydrolysis suppression typically involves coating AlN particles with a protective layer—via chemical bonding or physical adsorption—to isolate them from water.
Key methods include:
1. AlN Surface Modification——Surface Chemical Modification
This approach chemically reacts AlN particles with modifiers to form a passivation layer, enhancing surface properties.
Techniques include:
①Coupling agent modification
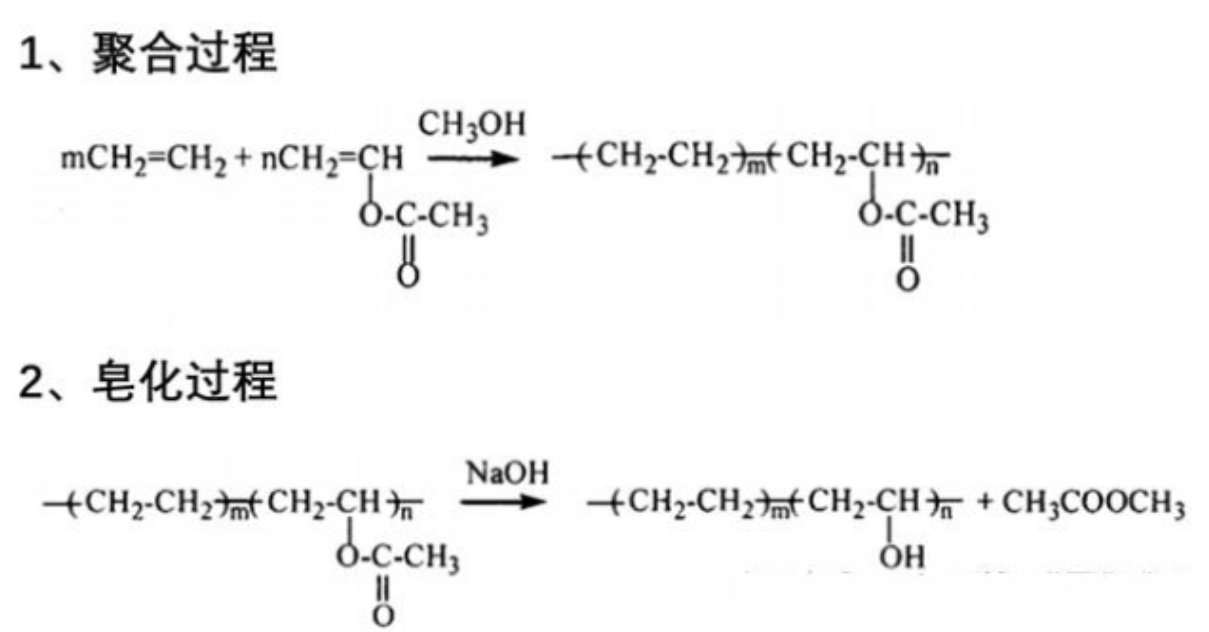
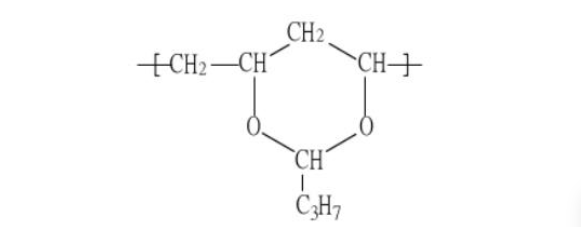
②Graft copolymerization modification
③Surface oxidation modification
④Surfactant modification

2. AlN Surface Treatment——Surface Physical Coating Treatment
(1) Liquid-Phase Coating
Modifiers are added to an AlN powder suspension, forming a coating via mechanical stirring. The coating adheres through adsorption or van der Waals forces, without chemical reactions.
(2) Vapor Deposition Coating
Utilizing the sublimation of materials (e.g., SiO), which are heated to deposit onto AlN particles, improving hydrolysis resistance. For example, SiO powder is sublimed in an Al₂O₃ crucible containing AlN powder, carbon felt, and graphite plates, forming a protective layer.
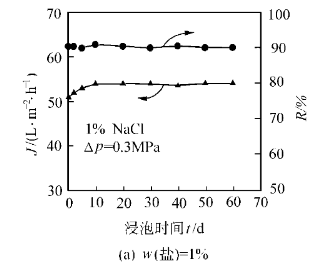
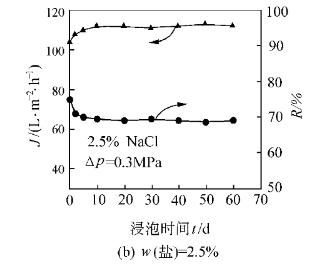
Alternatively, strong acid modifiers can be mixed with AlN powder via mechanical ball milling. This method avoids high temperatures, offers good reproducibility, and significantly enhances hydrolysis resistance while improving dispersion and stability in water—beneficial for preparing high-solid-content AlN ceramic slurries.

Summary
While all methods improve AlN powder’s hydrolysis resistance, each has limitations.
For instance:
Ceramic-grade AlN powder requires high purity. Silicon-based coatings (e.g., from silane) may introduce impurities during sintering, reducing thermal conductivity. Here, organic acids or thermal oxidation are preferred.
Thermal interface materials (TIMs) demand compatibility with silicone oils/gels for high filler loading and uniformity,Silane-based treatments are more suitable.
Combining multiple modification methods may yield superior results.

About Xiamen Juci Technology Co., Ltd.
Xiamen Juci Technology Co., Ltd. is a high-tech enterprise specializing in the research, development, production, and sales of high-performance AlN materials. As a leading AlN powder suppliers, the company is committed to providing high-quality aluminum nitride series products and solutions for industries such as electronics, semiconductors, and aerospace.Xiamen Juci Technology Co., Ltd. can effectively address the hydrolysis problem of AlN. With exceptional quality and service, Xiamen Juci has earned widespread trust from global customers.
Media Contact:
Xiamen Juci Technology Co., Ltd.
Phone: +86 592 7080230
Email: miki_huang@chinajuci.com
Website: www.jucialnglobal.com